Free ASVAB Practice Tests
-
Every Test is UniqueCustom software and unique templates randomize questions, answers, and variables every time you take a new test. You'll never take the same test twice!
-
1,557 Questions, Problems & Flash CardsHuge database of 668 multiple-choice questions, 135 math and algebra problems, and 754 flash cards to help you prepare for the ASVAB.
-
Detailed SolutionsGet a question wrong? All questions and problems have detailed answer explanations so you can learn exactly how to get it right the next time.
-
Know You're ReadyWant to know how you stack up? When you're done with a practice test you can compare your score to everyone else who has ever answered those questions.
-
Interactive Study GuideDetailed ASVAB study guide, MOS study guides, and line score study guides outline exactly what you should know to earn your target scores and customized tests and flash cards for each topic let you laser focus your limited study time.
-
Brand New for 2019ASVAB Test Bank has been completely redesigned for 2019 with all new questions, problems, and flash cards. And the redesign isn't done! Coming soon:
- More Content
- More questions, problems and flash cards
- Bookmarks
- Create a custom study guide with just the topics you're studying
- Score Estimator
- Custom estimate of your potential ASVAB score
Plus printable tests, Q&A, and an ad-free upgrade. Have a suggestion? Please let us know what you want!
Take an ASVAB Practice Test
Sample Practice Test Questions
The mechanical advantage of a wheel and axle is equal to the:
ratio of the diameters of the wheels
A wheel and axle uses two different diameter wheels mounted to a connecting axle. Force is applied to the larger wheel and large movements of this wheel result in small movements in the smaller wheel. Because a larger movement distance is being translated to a smaller distance, force is increased with a mechanical advantage equal to the ratio of the diameters of the wheels. An example of a wheel and axle is the steering wheel of a car.
Which of the following allows DC to pass easily but resists the flow of AC?
inductor
An inductor is coiled wire that stores electric energy in the form of magnetic energy and resists changes in the electric current flowing through it. If current is increasing, the inductor produces a voltage that slows the increase and, if current is decreasing, the magnetic energy in the coil opposes the decrease to keep the current flowing longer. In contrast to capacitors, inductors allow DC to pass easily but resist the flow of AC.
What part of the nervous system is responsible for controlling involuntary actions like breathing, swallowing, and heartbeat?
medulla
Part of the brainstem, the medulla is the connection between the brain and the spinal cord. It controls involuntary actions like breathing, swallowing, and heartbeat.

This tool is a(n) __________.
inside caliper
Calipers are similar to micrometers in shape but instead of measuring distances, calipers are used to transfer distances between objects. An outside caliper is used to transfer outside dimensions while an inside caliper is used to transfer inside distances. A vernier caliper is an extremely precise caliper (down to \({1 \over 1000}\) inch) that allows measuring / transferring either inside or outside dimensions.
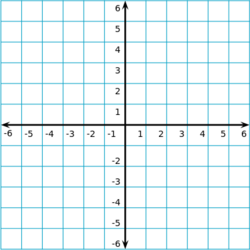
The endpoints of this line segment are at (-2, -7) and (2, 3). What is the slope of this line?
The slope of this line is the change in y divided by the change in x. The endpoints of this line segment are at (-2, -7) and (2, 3) so the slope becomes:
m = \( \frac{\Delta y}{\Delta x} \) = \( \frac{(3.0) - (-7.0)}{(2) - (-2)} \) = \( \frac{10}{4} \)m = 2\(\frac{1}{2}\)
If an electrical circuit is interrupted, which of the following will result?
open circuit
A closed circuit is a complete loop or path that electricity follows. It consists of a source of voltage, a load, and connective conductors. If the circuit is interrupted, if a wire is disconnected or cut for example, it becomes an open circuit and no electricity will flow.
What is a framing square used for?
marking lines at a right angle
Rulers are used to both measure distance and to draw straight lines. Tape measures are flexible rulers made from cloth or metal and are useful for measuring longer distances and in tighter spaces. Both rulers and tape measures provide similar accuracy, commonly down to \({1 \over 16}\) or \({1 \over 32}\) inch. A steel framing square is made up of a shorter ruler (tongue) and a longer ruler (blade) that meet at a 90° (right) angle. A framing square is often used by carpenters when framing stairs and roofs.
In cell biology, the primary difference between anaerobic and aerobic respiration is that aerobic respiration requires:
oxygen
Some plant cells produce their own energy through photosynthesis which is the process by which sunlight, carbon dioxide, and water react to make sugar and oxygen. Animal cells cannot produce their own energy and, instead, generate energy when mitochondria consume outside sugar and oxygen through aerobic respiration.